2,2-DI-N-PROPYLACETYL CHLORIDE
Propylvaleroylchloride
CAS: 2936-08-5
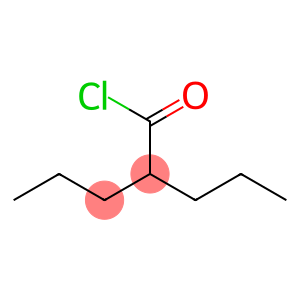
Molecular Formula: C8H15ClO
2,2-DI-N-PROPYLACETYL CHLORIDE - Names and Identifiers
| Name | Propylvaleroylchloride |
| Synonyms | VALPROYL CHLORIDE Propylvaleroylchloride 2-PROPYLVALERYL CHLORIDE 2-propylpentanoyl chloride DI-N-PROPYLACETYL CHLORIDE 2-N-Propylpentanoylchloride 2,2-DI-N-PROPYLACETYL CHLORIDE 2-n-Propyl-n-valeroyl chloride 2,2-Di-n-propylacetyl chloride 2-N-PROPYL-N-VALEROYL CHLORIDE Heptane-4-carbonyl chloride~2-n-Propylpentanoyl chloride~2-n-Propylvaleryl chloride |
| CAS | 2936-08-5 |
| EINECS | 220-914-5 |
| InChI | InChI=1/C8H15ClO/c1-3-5-7(6-4-2)8(9)10/h7H,3-6H2,1-2H3 |
2,2-DI-N-PROPYLACETYL CHLORIDE - Physico-chemical Properties
| Molecular Formula | C8H15ClO |
| Molar Mass | 162.66 |
| Density | 0,95 g/cm3 |
| Boling Point | 68-70°C 10mm |
| Flash Point | 68-70°C/10mm |
| Water Solubility | Reacts with water. |
| Vapor Presure | 0.739mmHg at 25°C |
| Appearance | clear liquid |
| Color | Colorless to Light yellow |
| BRN | 1098856 |
| Storage Condition | Room Temprature |
| Sensitive | Moisture Sensitive |
| Refractive Index | 1.4310-1.4330 |
| MDL | MFCD00051446 |
2,2-DI-N-PROPYLACETYL CHLORIDE - Risk and Safety
| Risk Codes | R34 - Causes burns R22 - Harmful if swallowed |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | 3265 |
| Hazard Note | Corrosive |
| Hazard Class | 8 |
| Packing Group | II |
2,2-DI-N-PROPYLACETYL CHLORIDE - Introduction
Propylvaleroylchloride is an organic compound with the chemical formula C9H17ClO. It is a colorless to pale yellow liquid with a chilli smell and pungent odor.
Propylvaleroylchloride is commonly used as an acylating reagent in chemical synthesis. It can react with alcohols, phenols, amines, etc. to generate the corresponding esters, ketones and amides. Therefore, it has a wide range of applications in the pharmaceutical, pesticide and other fields. In addition, it can also be used as an intermediate for organic synthesis reactions.
The method for preparing Propylvaleroylchloride is usually obtained by reacting 2,2-di-n-propyl acetic acid with thionyl chloride. The specific steps are as follows:
1. 2,2-di-n-propyl acetic acid and thionyl chloride are dissolved in the reaction solvent, such as chlorinated hydrocarbons or ethers.
2. Slowly add the sulfoxide solution Dropwise to the solution of 2,2-di-n-propyl acetic acid at low temperature, while adding the acid catalyst.
3. After completion of the reaction, the reaction mixture was neutralized with water to obtain Propylvaleroylchloride.
It should be noted that the operating conditions and addition speed should be paid attention to during the preparation process to avoid violent reaction or hydrolysis.
Regarding the safety information of Propylvaleroylchloride, it is an irritating compound that may cause skin and eye irritation after contact. During the operation, it is necessary to avoid contact with skin, eyes and respiratory tract. In case of accidental contact, rinse immediately with plenty of water and seek medical help. At the same time, it should be operated in a well-ventilated place to avoid inhaling its vapor. In addition, it is also a flammable liquid, should be away from fire and high temperature environment.
Propylvaleroylchloride is commonly used as an acylating reagent in chemical synthesis. It can react with alcohols, phenols, amines, etc. to generate the corresponding esters, ketones and amides. Therefore, it has a wide range of applications in the pharmaceutical, pesticide and other fields. In addition, it can also be used as an intermediate for organic synthesis reactions.
The method for preparing Propylvaleroylchloride is usually obtained by reacting 2,2-di-n-propyl acetic acid with thionyl chloride. The specific steps are as follows:
1. 2,2-di-n-propyl acetic acid and thionyl chloride are dissolved in the reaction solvent, such as chlorinated hydrocarbons or ethers.
2. Slowly add the sulfoxide solution Dropwise to the solution of 2,2-di-n-propyl acetic acid at low temperature, while adding the acid catalyst.
3. After completion of the reaction, the reaction mixture was neutralized with water to obtain Propylvaleroylchloride.
It should be noted that the operating conditions and addition speed should be paid attention to during the preparation process to avoid violent reaction or hydrolysis.
Regarding the safety information of Propylvaleroylchloride, it is an irritating compound that may cause skin and eye irritation after contact. During the operation, it is necessary to avoid contact with skin, eyes and respiratory tract. In case of accidental contact, rinse immediately with plenty of water and seek medical help. At the same time, it should be operated in a well-ventilated place to avoid inhaling its vapor. In addition, it is also a flammable liquid, should be away from fire and high temperature environment.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 2,2-Di-n-butyl-1,3-propanediol Request for quotation
CAS: 2936-08-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 2936-08-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-Propylvaleroyl Chloride Visit Supplier Webpage Request for quotation
CAS: 2936-08-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 2936-08-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2,2-Di-n-butyl-1,3-propanediol Request for quotation
CAS: 2936-08-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 2936-08-5
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-Propylvaleroyl Chloride Visit Supplier Webpage Request for quotation
CAS: 2936-08-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 2936-08-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

